 The MHRA Orange Guide
The MHRA Orange Guide
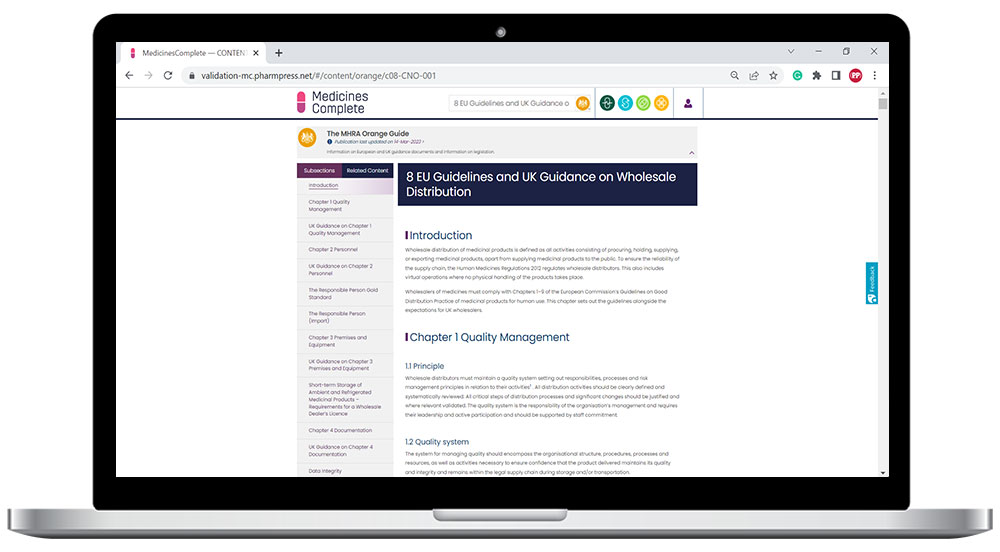

The reference source for manufacturers and distributors of medicines
Based on current regulation, and regularly updated to reflect changes in best practices, legal requirements, and emerging technologies.
Rules and Guidance for Pharmaceutical Manufacturers and Distributors (The Orange Guide) is compiled by the Medicines and Healthcare Regulatory Agency (MHRA) as the single source of European and UK guidance and UK legislation on the manufacture and distribution of human medicines, active substances, and brokering medicines.
Supports medicines manufacturers and distributors in meeting legal requirements

The Code of Practice for Qualified Persons

Conditions and guidance for investigational medicinal products

Incorporates changes made after the UK’s exit from the European Union

Minimises research time

Aids good practice

GMP and GDP guidance for active substances

Naming of sites on a licence

Provides a framework for continuous improvement in quality control and monitoring
Request access today
Access The MHRA Orange Guide through MedicinesComplete today.
Contact us now for pricing and access information.
Related publications
-
 FEATURED
FEATUREDHandbook of Pharmaceutical Excipients – Ninth Edition
Guide to the properties for excipients, their safe use and application.
Info -
 FEATURED
FEATUREDDrugs in Use: Case Studies for Pharmacists and Prescribers Sixth Edition
Bridges the gap between theoretical knowledge about medicines and its practical application to patie…
Info -
 PRE-ORDER
PRE-ORDERBNF for Children (BNFC) 2022-2023
The first choice for concise medicines information for children.
Info
See all our printed publications in the Shop.





